Well, if I could actually hear your responses, I would hope that the answer would be, "Yes! Go for it!" So without further ado:
Here is a Haiku about why and how atoms form covalent bonds by sharing electrons:
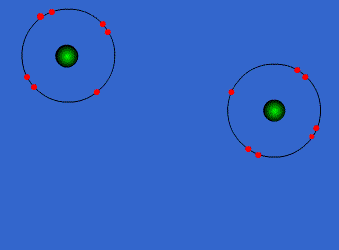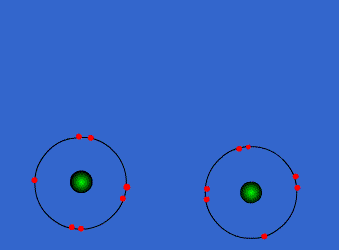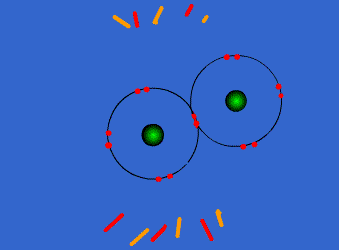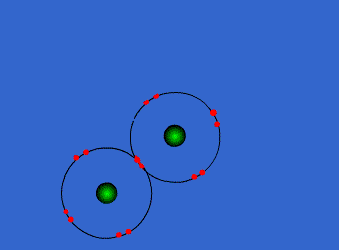
Because the electron configurations of noble gasses (8 valence electrons and completely full outer shells) are the most stable of all configurations, when forming compounds, atoms strive to gain an octet. Thus, they will share the number of electrons needed for all atoms to have eight electrons in their highest energy level shells.
The image to the left shows how two Group 17 (7A) elements (two atoms with 7 valence electrons) can combine to create one single bond and a diatomic molecule.
Works Cited:
Background Image for Haiku: http://205.243.100.155/frames/lichtenbergs.html
Animated .gif: http://teachers.pasd.k12.pa.us/hs/plavcand/Ionic%20and%20Covalent%20Bonds.htm
----//---- ----//----
Now, here is a short Animoto (an juiced up slide show) about five or six of the simplest molecular geometries and some household items that resemble these shapes. Enjoy!!
Works Cited:
See last slide of movie.

Wow Chris.....awesome video...I am a chem teacher and I plan on sharing this video with my students!
ReplyDeleteHi Chris,
ReplyDeleteAs an assignment for an online class I am taking, I had to find a student blog that impressed me. Needless to say, I chose yours! I am very impressed by your use of video, the color, and the 3D electron affinity chart. I also like the chemical equation balancer at the bottom of your blog which I’m sure many chemistry students find useful. In my own classroom, I could see asking students to create a Chemistry (or Math, or Biology blog.) The students would need to post once a week as an assignment. The post could be a video, a chart, or picture but it would have to be something that related to one of the topics being taught that week. Thanks for such an inspiring blog! Morisa
Hi Chris,
ReplyDeleteI forgot to include a link to my comment about you on my blog...it's http://mathmompa.wordpress.com/2011/02/26/student-blogging/
Thanks again! Morisa
Hey, great Haiku. And people say chemistry can't be fun... Shame on them! Great site, by the way.
ReplyDeleteThank you for this thoughtful post.. it is helpful & your postmotivated me..thank
ReplyDeleteOnline Diploma
Its first wish of every student to get good marks in SAT test but problems is that which Math tutor books are Good for test preparation and helpful for getting good grades. And some perfect books give the Best results.
ReplyDeleteI really like your take on the issue. I now have a clear idea on what this matter is all about.. fake bachelor degree
ReplyDeleteI am glad that I saw this post. It is informative blog for us and we need this type of blog thanks for share this blog, Keep posting such instructional blogs and I am looking forward for your future posts.samsung un82mu8000
ReplyDeleteWe also provide analytical services and laboratory services to our customers. Vat Yellow 2
ReplyDeleteThis is my first time i visit here. I found so many interesting stuff in your blog especially its discussion. From the tons of comments on your articles, I guess I am not the only one having all the enjoyment here keep up the good work Science
ReplyDeleteI am thankful to this blog for assisting me. I added some specified clues which are really important for me to use them in my writing skill. Really helpful stuff made by this blog.Emerson Liebert GXT3 On Line
ReplyDeleteMy own perspectives are coordinating with creator and I have encountered such. Your blog is extremely instructive. study material for distance learning courses Chem Academy offering distance learning courses for IIT JAM and best Correspondence course for IIT JAM. We also provide CSIR UGC NET, BHU , TIFR , JNU and DU Entrance Exam distance learning courses .
ReplyDeleteIt was truly decent to peruse article composed on this blog. I might likewise want to include couple of strategies with best of my insight which can help peruser to an ever increasing extent. CSIR NET Coaching in Jaipur
ReplyDeleteIntroduction to online course for IIT-JAM, CSIR-NET and GATE | Chem Introduction to online course for IIT-JAM, CSIR-NET and GATE | You tube mirage vs complete Online course https://www.youtube.com/watch?v=8paWv4BIzxA
ReplyDeleteThe intensity of articles can with out issues be felt of this weblog. Very specific and without delay to the mark. I understood with out difficulty the problem of fact which the author of this weblog wanted to deliver thru his mind. looking for more. Online Test series for iit jam physics
ReplyDelete